PRODUCTS
NUGARD
- Technical description
- Dimensions and Tolerances
- Guidelines for the correct use and application of the product
- Precautions, restrictions, warnings and special care
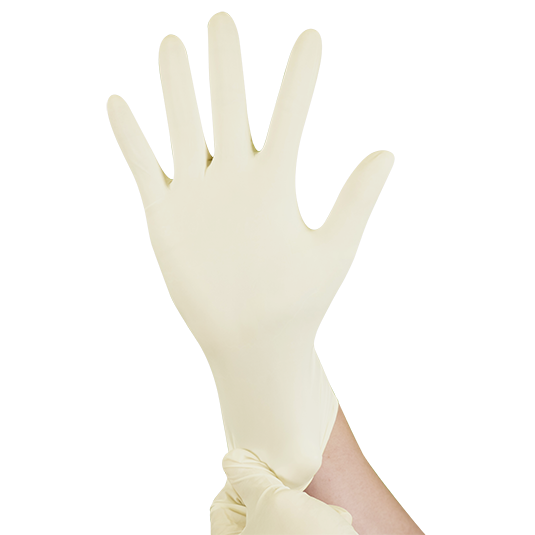
Procedural glove, non-sterile, made of natural latex, ambidextrous, lightly lubricated with non-toxic bioabsorbable powder (starch USP grade modified corn).
Composition: 100% natural rubber latex.
| SIZES | COMMERCIAL BOX | TRANSPORT BOX | WEIGHT | LARGE | ALT | COMP | BAR CODE |
| PP | 100 units | 1.000 units | 5,3 | 0,23 | 0,23 | 0,33 | 7898994 049814 |
| P | 100 units | 1.000 units | 5,9 | 0,23 | 0,23 | 0,33 | 7898994 049821 |
| M | 100 units | 1.000 units | 6,2 | 0,23 | 0,23 | 0,33 | 7898994 049838 |
| G | 100 units | 1.000 units | 7,1 | 0,23 | 0,23 | 0,33 | 7898994 049845 |
| SIZE | WIDTH (mm) | LENGTH (mm) | SMOOTH AREA THICKNESS (MM) | CENTER APPROXIMATE OF PALMA AREA LISA |
| PP | ≤ 80 | 220 | ≥ 0,08 | ≤ 2,00 |
| P | 80 ± 10 mm | 220 | ≥ 0,08 | ≤ 2,00 |
| M | 95 ± 10 mm | 230 | ≥ 0,08 | ≤ 2,00 |
| G | 110 ± 10 mm | 230 | ≥ 0,08 | ≤ 2,00 |
For correct use of the product, a brief asepsis must be done first, with hand washing and subsequent drying, so that the gloves are then put on, before activities by professionals exposed to the risk of contamination.
- Contains natural latex, may cause allergy;
- There are isolated cases of allergic reactions to latex;
- If there is skin irritation, stop using the material and seek medical advice;
- Do not use if the packaging is violated;
- Single use product – Never reuse the glove; Destroy after use;
- Discard the glove whenever there is doubt about the contamination;
- Observe o tamanho da luva antes do uso para evitar que a mesma se rasgue;
- Store in a cool, dry place at room temperature.
Classification: NUGARD procedure gloves meet the INMETRO Ordinances 332 and 451, RDC 55/2011 Glove for non-surgical procedure, ABNT NBR ISO 11193-1 Single-Use Medical Examination Gloves, Part 1: Specification for Gloves Produced from Rubber Latex or Rubber Solution and NR6 of the MINISTRY OF LABOR.
Commercial Name: Latex Procedure Glove – With Powder
Certificate of conformity – INMETRO CE-LVL/ICEPEX-I 00063-33-1
Approval certificate – CA :40.389
Registry ANVISA: 80748910009 – Made in Malaysia